Навигация
Educational institution
Summary
Subject: Francium
He performed the: .
Checked: .
City: 20 г.
Content
1 Introduction
2 Characteristics
3 Applications
4 History
4.1 Erroneous and incomplete discoveries
4.2 Perey's analysis
5 Occurrence
5.1 Natural
5.2 Synthesized
6 Isotopes
7 Conclusion
8 References
1. Introduction
Francium (pronounced /ˈfrænsiəm/, FRAN-see-əm), formerly known as eka-caesium and actinium K,[1] is a chemical element that has the symbol Fr and atomic number 87. It has one of the lowest electronegativity of all known elements, and is the second rarest naturally occurring element (after astatine). Francium is a highly radioactive metal that decays into astatine, radium, and radon. As an alkali metal, it has one valence electron. Francium was discovered by Marguerite Perey in France (from which the element takes its name) in 1939. It was the last element discovered in nature, rather than synthesized.[2] Outside the laboratory, francium is extremely rare, with trace amounts found in uranium and thorium ores, where the isotope francium-223 continually forms and decays. As little as 20-30 g (one ounce) exists at any given time throughout the Earth's crust; the other isotopes are entirely synthetic. The largest amount ever collected of any isotope was a cluster of about 10,000 atoms (of francium-210) created as an ultracold gas at Stony Brook in 1997.[3] General properties
Name, symbol, number francium, Fr, 87
Element category alkali metal
Group, period, block 1, 7, s
Standard atomic weight (223) g·mol−1
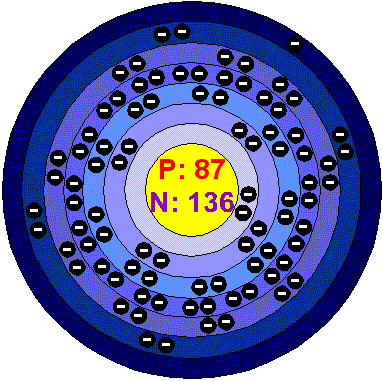
Electron configuration [Rn] 7s1
Electrons per shell 2, 8, 18, 32, 18, 8, 1 (Image)
Physical properties
Phase solid
Density (near r.t.) 1.87 g·cm−3
Melting point ? 300 K, ? 27 °C, ? 80 °F
Boiling point ? 950 K, ? 677 °C, ? 1250 °F
Heat of fusion ca. 2 kJ·mol−1
Heat of vaporization ca. 65 kJ·mol−1
2. Characteristics
Francium is the least stable of the naturally occurring elements: its most stable isotope, francium-223, has a maximum half-life of only 22 minutes. In contrast, astatine, the second-least stable naturally occurring element, has a maximum half-life of 8.5 hours.[4] All isotopes of francium decay into either astatine, radium, or radon.[4] Francium is also less stable than all synthetic elements up to element 105.[3]
Francium is an alkali metal whose chemical properties most resemble those of caesium.[3] A very heavy element with a single valence electron,[5] it has the highest equivalent weight of any element.[3] Liquid francium — if such a substance were to be created — should have a surface tension of 0.05092 N/m at its melting point.[6] Francium’s melting point was claimed to have been calculated to be around 27 °C (80 °F, 300 K). However, the melting point is uncertain because of the element’s extreme rarity and radioactivity. This melting point may have been in limited precision, or so much heat produced from radioactivity that its calculated melting point may have been overestimated. However, the melting point of francium is estimated to be about 22 °C (71 °F, 295 K), based from the periodic trends in melting points with other alkali metals.[citation needed] Also the boiling point may have been overestimated at around 677 °C (1250 °F, 950 K). Based from the periodic trends with other alkali metals, the boiling point of francium is estimated to be between 660 to 665 °C (1220 to 1230 °F, 935 to 940 K).[citation needed] Because radioactive elements give off heat Francium would almost certainly be a liquid if enough visible Francium were to be produced.
Linus Pauling estimated the electronegativity of francium at 0.7 on the Pauling scale, the same as caesium;[7] the value for caesium has since been refined to 0.79, although there are no experimental data to allow a refinement of the value for francium.[8] Francium has a slightly higher ionization energy than caesium,[9] 392.811(4) kJ/mol as opposed to 375.7041(2) kJ/mol for caesium, as would be expected from relativistic effects, and this would imply that caesium is the less electronegative of the two.
Francium coprecipitates with several caesium salts, such as caesium perchlorate, which results in small amounts of francium perchlorate. This coprecipitation can be used to isolate francium, by adapting the radiocaesium coprecipitation method of Glendenin and Nelson. It will additionally coprecipitate with many other caesium salts, including the iodate, the picrate, the tartrate (also rubidium tartrate), the chloroplatinate, and the silicotungstate. It also coprecipitates with silicotungstic acid, and with perchloric acid, without another alkali metal as a carrier, which provides other methods of separation.[10][11] Nearly all francium salts are water-soluble.[12]
Похожие работы
... не дає настільки точних результатів, як сучасні способи виміру температури. Звідси і розбіжності, що залежать від того, які теоретичні передумови покладені в основу розрахунку. Розділ 4. Хімічні властивості Теоретично францій повинен бути практично повним хімічним аналогом своїх найближчих сусідів по групі лужних металів – цезію та рубідію. Зокрема, його ступінь окислення у сполуках завжди ...
0 комментариев