Навигация
3. Applications
Due to its instability and rarity, there are no commercial applications for francium.[13][14][15][16][17] It has been used for research purposes in the fields of biology [18] and of atomic structure. Its use as a potential diagnostic aid for various cancers has also been explored,[4] but this application has been deemed impractical.[15]
Francium's ability to be synthesized, trapped, and cooled, along with its relatively simple atomic structure have made it the subject of specialized spectroscopy experiments. These experiments have led to more specific information regarding energy levels and the coupling constants between subatomic particles.[19] Studies on the light emitted by laser-trapped francium-210 ions have provided accurate data on transitions between atomic energy levels which are fairly similar to those predicted by quantum theory.[20]
4. History
As early as 1870, chemists thought that there should be an alkali metal beyond caesium, with an atomic number of 87.[4] It was then referred to by the provisional name eka-caesium.[21] Research teams attempted to locate and isolate this missing element, and at least four false claims were made that the element had been found before an authentic discovery was made.
4.1 Erroneous and incomplete discoveries
Soviet chemist D.K. Dobroserdov was the first scientist to claim to have found eka-caesium, or francium. In 1925, he observed weak radioactivity in a sample of potassium, another alkali metal, and concluded that eka-caesium was contaminating the sample.[22] He then published a thesis on his predictions of the properties of eka-caesium, in which he named the element russium after his home country.[23] Shortly thereafter, Dobroserdov began to focus on his teaching career at the Polytechnic Institute of Odessa, and he did not pursue the element further.[22]
The following year, English chemists Gerald J. F. Druce and Frederick H. Loring analyzed X-ray photographs of manganese(II) sulfate.[23] They observed spectral lines which they presumed to be of eka-caesium. They announced their discovery of element 87 and proposed the name alkalinium, as it would be the heaviest alkali metal.[22]
In 1930, Fred Allison of the Alabama Polytechnic Institute claimed to have discovered element 87 when analyzing pollucite and lepidolite using his magneto-optical machine. Allison requested that it be named virginium after his home state of Virginia, along with the symbols Vi and Vm.[23][24] In 1934, however, H.G. MacPherson of UC Berkeley disproved the effectiveness of Allison's device and the validity of this false discovery.[25]
In 1936, Romanian chemist Horia Hulubei and his French colleague Yvette Cauchois also analyzed pollucite, this time using their high-resolution X-ray apparatus.[22] They observed several weak emission lines, which they presumed to be those of element 87. Hulubei and Cauchois reported their discovery and proposed the name moldavium, along with the symbol Ml, after Moldavia, the Romanian province where they conducted their work.[23] In 1937, Hulubei's work was criticized by American physicist F. H. Hirsh Jr., who rejected Hulubei's research methods. Hirsh was certain that eka-caesium would not be found in nature, and that Hulubei had instead observed mercury or bismuth X-ray lines. Hulubei, however, insisted that his X-ray apparatus and methods were too accurate to make such a mistake. Because of this, Jean Baptiste Perrin, Nobel Prize winner and Hulubei's mentor, endorsed moldavium as the true eka-caesium over Marguerite Perey's recently discovered francium. Perey, however, continuously criticized Hulubei's work until she was credited as the sole discoverer of element 87.[22]
4.2 Perey's analysis
Eka-caesium was discovered in 1939 by Marguerite Perey of the Curie Institute in Paris, France when she purified a sample of actinium-227 which had been reported to have a decay energy of 220 keV. However, Perey noticed decay particles with an energy level below 80 keV. Perey thought this decay activity might have been caused by a previously unidentified decay product, one which was separated during purification, but emerged again out of the pure actinium-227. Various tests eliminated the possibility of the unknown element being thorium, radium, lead, bismuth, or thallium. The new product exhibited chemical properties of an alkali metal (such as coprecipitating with caesium salts), which led Perey to believe that it was element 87, caused by the alpha decay of actinium-227.[21] Perey then attempted to determine the proportion of beta decay to alpha decay in actinium-227. Her first test put the alpha branching at 0.6%, a figure which she later revised to 1%.[26]
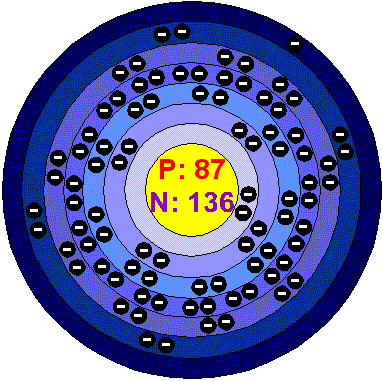
Perey named the new isotope actinium-K (now referred to as francium-223)[21] and in 1946, she proposed the name catium for her newly discovered element, as she believed it to be the most electropositive cation of the elements. Irène Joliot-Curie, one of Perey's supervisors, opposed the name due to its connotation of cat rather than cation.[21] Perey then suggested francium, after France. This name was officially adopted by the International Union of Pure and Applied Chemistry in 1949,[4] becoming the second element after gallium to be named after France. It was assigned the symbol Fa, but this abbreviation was revised to the current Fr shortly thereafter.[27] Francium was the last element discovered in nature, rather than synthesized, following rhenium in 1925.[21] Further research into francium's structure was carried out by, among others, Sylvain Lieberman and his team at CERN in the 1970s and 1980s.[28]
5. Occurrence
5.1 Natural
Francium-223 is the result of the alpha decay of actinium-227 and can be found in trace amounts in uranium and thorium minerals.[3] In a given sample of uranium, there is estimated to be only one francium atom for every 1×1018 uranium atoms.[15] It is also calculated that there is at most 30 g of francium in the earth's crust at any time.[29] This makes it the second rarest element in the crust after astatine.[4][15]

Figure. 0. This sample of uraninite contains about 100,000 atoms (3.3 ? 10?20 g) of 223Fr at any given time.[15]
Похожие работы
... не дає настільки точних результатів, як сучасні способи виміру температури. Звідси і розбіжності, що залежать від того, які теоретичні передумови покладені в основу розрахунку. Розділ 4. Хімічні властивості Теоретично францій повинен бути практично повним хімічним аналогом своїх найближчих сусідів по групі лужних металів – цезію та рубідію. Зокрема, його ступінь окислення у сполуках завжди ...
0 комментариев